Life is organized at many length scales ranging from the molecular to the macro, and there is an incredible amount of complexity in the morphologies at these various levels of organization. Historically, biologists have looked to the genetic code for instructions to reveal the secrets of self-assembly. However, once DNA is transcribed and translated to proteins and subsequent other macromolecules, there is no known “code” that dictates how these products interact. To discern the principles of this self-organization, we must identify the physical phenomena that govern the soft, disordered, and often far-from-equilibrium living world. I elucidate the “rules of living patterns” that lead to a variety of shapes and add to our understanding of how life builds itself.
Liquid-liquid phase separation leads to pollen wall pattern formation
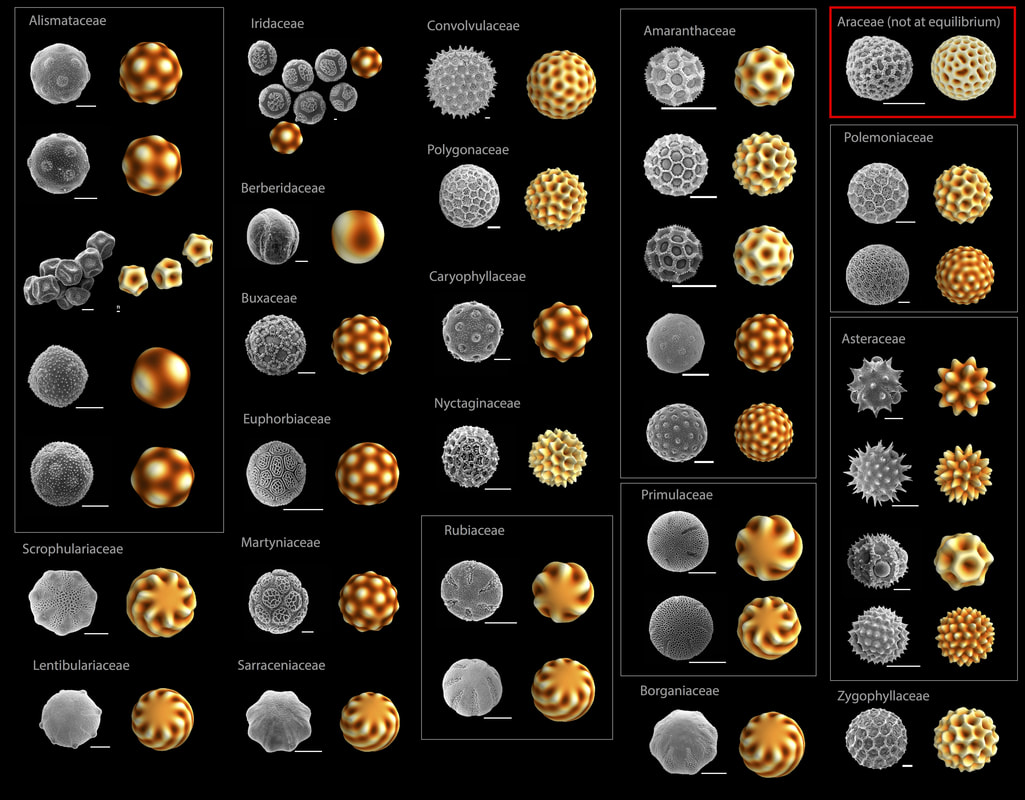
Pollen grains are known for their impressive variety of species-specific, microscale surface patterning. Despite having similar biological developmental steps, pollen grain surface features are remarkably geometrically varied. Previous work suggests that a physical process may drive this pattern formation and that the observed diversity of patterns can be explained by viewing pollen pattern development as a phase transition to a spatially modulated phase. Several studies have shown that the polysaccharide material of plant cell walls undergoes phase separation in the absence of cross-linking stabilizers of the mixed phase. Here we show experimental evidence that phase separation of the extracellular polysaccharide material (primexine) during pollen cell development leads to a spatially modulated phase. The spatial pattern of this phase-separated primexine is also mechanically coupled to the undulation of the pollen cell membrane. The resulting patterned pools of denser primexine form the negative template of the ultimate sites of sporopollenin deposition, leading to the final micropattern observed in the mature pollen. We then present a general physical model of pattern formation via modulated phases. Using analytical and numerical techniques, we find that most of the pollen micropatterns observed in biological evolution could result from a physical process of modulated phases. However, an analysis of the relative rates of transitions from states that are equilibrated to or from states that are not equilibrated suggests that while equilibrium states of this process have occurred throughout evolutionary history, there has been no particular evolutionary selection for symmetric, equilibrated states.
Publications related to this project:
Radja A, Horsley EM, Lavrentovich MO, and Sweeney AM. (2019). Pollen cell wall patterns form from modulated phases. Cell. In Press.
Lavrentovich MO, Horsley EM, Radja A, Sweeney AM, and Kamien RD. (2016). First-order patterning transitions on a sphere as a route to cell morphology. PNAS. 113 (19) 5189-5194.
Liu JQ, Radja A, Gao Y, Yin R, Sweeney AM, and Yang S. (2020). Mimicry of a biophysical pathway leads to diverse pollen-like surface patterns. PNAS.
Biological structural networks
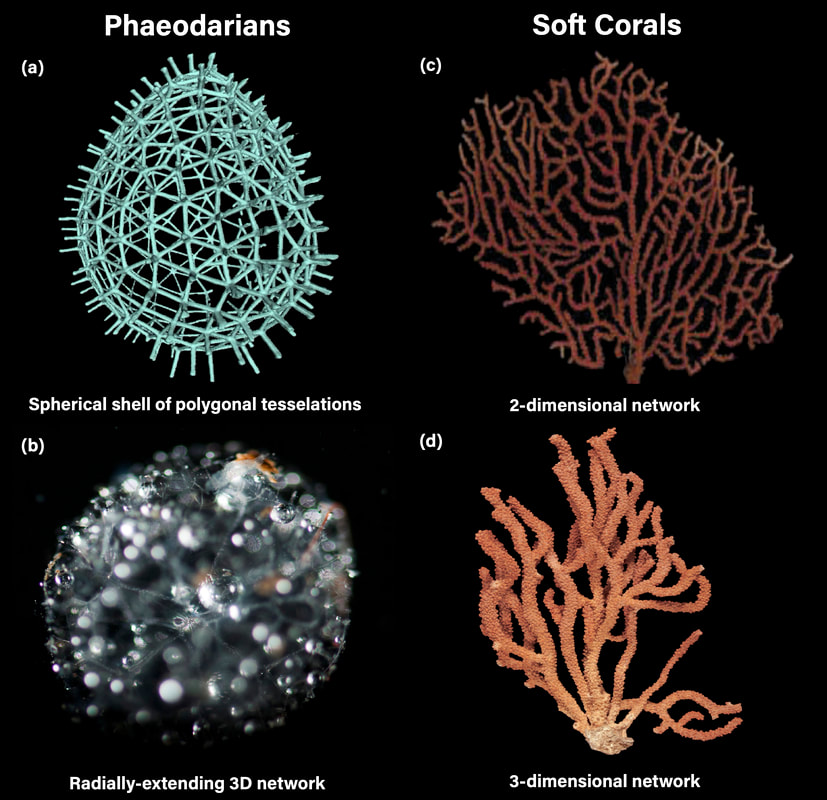
(a, b) Radja, (c) (Breedy and Guzman, 2018), (d) (Horvath, 2019)
Another common but relatively understudied pattern found in living systems is structural networks. Branching biological structures can be observed across taxa, from airways and blood vessels to root systems and slime mold tissues . Despite their ubiquity, an understanding of both useful quantification metrics and pattern formation mechanisms is lacking in biological structural networks. I am studying two marine organisms with some of the most diverse and geometrically-intricate network structures seen in nature (satisfying the reductionist system requirements outlined on page 1): (a) the spherical networks of silica skeletons in deep-sea single-celled protists called Phaeodarians and (b) the 2/3D tree-like tissue networks of soft corals. The insights gained from these studies will help determine the physical principles that living systems have harnessed to create a diverse set of morphologies.